25 Jan 2024
The story behind the perfect chocolate
The control of the crystal structure V's nucleation, growth, and stability in the polymorphic system, which is so close and easy to transform each other, has become the focus and difficulty of chocolate production and storage, as well as the research focus of academic experts. As the saying goes, once a single link goes wrong, it is hard to maintain the perfection of chocolate.
Tempering technique
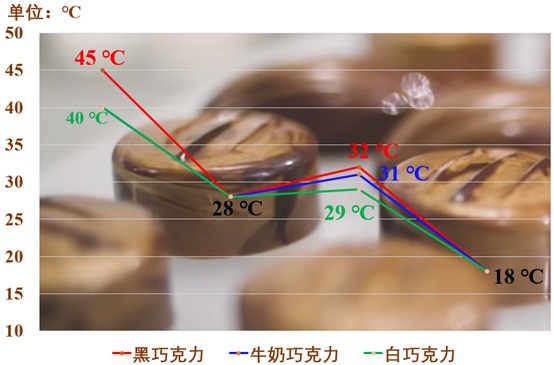
Temperature plays a critical role in crystal nucleation/growth and stability, especially in chocolate production. Achieving the desired crystal types is crucial, making it necessary to employ formulation development services that can offer accurate temperature control throughout the production process. The traditional tempering process is widely used to produce "perfect" chocolate: first, the chocolate needs to be heated to above the melting point threshold of all crystal types, which is about 45 ℃ (the melting point of chocolate will decrease accordingly after adding fat content, such as milk chocolate, white chocolate, etc., and the temperature needs to be appropriately reduced), making it lose all crystalline forms and become a molten state; then, cool it down to 28 ℃ (the nucleation temperature of V type, which is conducive to the nucleation and growth of a large number of V types. If the temperature is too low, it will cause more unstable crystal types to nucleate and grow, and the transformation of unstable crystal types into V types is not a one-time event, which will undoubtedly prolong the cooling process and cannot control the main crystal type composition in the finished product); then, warm it up to 32 ℃ to promote the nucleation and growth of V types, and more importantly, convert the unstable crystal types into V types to achieve the controllability of chocolate crystal types; finally, molding, cooling, and packaging.
In addition, external force shear needs to be added in the tempering process, such as hand flipping and cutting or using a tempering machine for commercial products like the one shown in the figure below. The shearing action can not only reduce the grain size during the cooling process and make it evenly distributed to increase the number of crystal seeds; but also promote mass and heat transfer during the entire tempering process, increasing the rate of unstable crystal types transforming into V types.
After reading this, do you think you can make chocolate too? Then be sure to control the temperature during the process. Secondly, you need to have a smooth marble board and flip and cut the chocolate quickly on it (this is a physical activity, so you also need strong arms~).
Chocolate "bloom"
For a long time, the deterioration of food has always been a focus of people's attention. Unlike other foods, the "deterioration" of chocolate is not decay, but rather a hardening and roughening of its texture, and a white film may appear on its surface, which looks like it is moldy. This is the so-called "bloom" of chocolate.

The chocolate produced by tempering is more stable and less likely to bloom, but improper storage or too long storage time can cause blooming. This white film has two forms, sugar bloom, and fat bloom. Sugar bloom is due to sugar in the chocolate dissolving in the condensed water on its surface, which evaporates and separates, causing the chocolate surface to become rough and granular. Fat bloom is due to fat substances precipitating on the surface of the chocolate, causing it to lose its luster and possibly causing a transformation of crystal types resulting in a hardening texture. By applying crystallization development techniques, we can minimize these issues and hopefully enjoy perfectly crafted chocolate free from both sugar and fat bloom.
Storing chocolate in a low-humidity, light-proof, and constant temperature environment can effectively prevent sugar from blooming; but fat bloom may involve the transition of metastable crystal type V to stable crystal type VI, which is a thermodynamically stable process. This seems to be verified by Wille's and Lutton's team in their findings: they found that almost all the cocoa butter crystal types of blooming chocolate were type VI. At the same time, it has been experimentally proved that VI types cannot be directly crystallized from the molten state of chocolate and must be transformed from the V type at a higher temperature. Although they cannot provide experimental data to prove that the reason for the blooming of chocolate is due to the transformation of V type to VI type, this conclusion can be inferred: temperature changes cause lipid molecules in chocolate components to migrate to the surface, precipitate and crystallize, and cause the VI type of cocoa butter to grow from a microscopic state to a macroscopic state.