News
13 Nov 2024
Improving the Manufacture of mRNA Biologics
13 Nov 2024
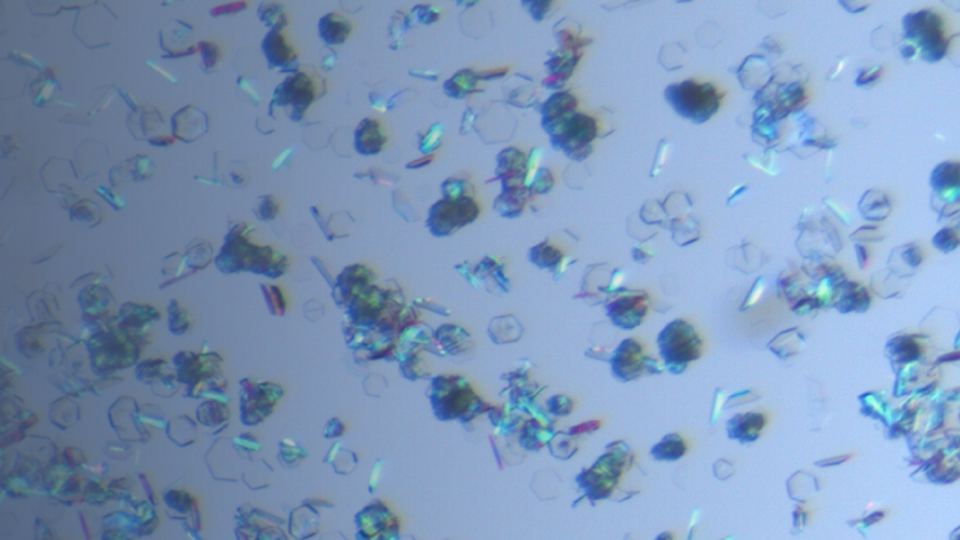 Amorphous Solid Dispersions
Amorphous solid dispersions have been shown to alter the properties of an API, including solubility, dissolution, and bioavailability. A variety of polymers or additives are available, and screening i...
Amorphous Solid Dispersions
Amorphous solid dispersions have been shown to alter the properties of an API, including solubility, dissolution, and bioavailability. A variety of polymers or additives are available, and screening i...
 Crystal Pharmatech Returned to AAPS 2022 PharmSci 360
Crystal Pharmatech returned to the American Association of Pharmaceutical Scientists (AAPS) 2022 PharmSci 360
conference alongside pharmaceutical industry players, buyers, and suppliers.
Crystal Pharmatech Returned to AAPS 2022 PharmSci 360
Crystal Pharmatech returned to the American Association of Pharmaceutical Scientists (AAPS) 2022 PharmSci 360
conference alongside pharmaceutical industry players, buyers, and suppliers.
 Fun Sharing: The Secret Behind the Silky Smoothness of Chocolate (Part 2)
The story behind the perfect chocolateThe control of the crystal structure V's nucleation, growth, and stability in the polymorphic system, which is so close and easy to transform each other, has ...
Fun Sharing: The Secret Behind the Silky Smoothness of Chocolate (Part 2)
The story behind the perfect chocolateThe control of the crystal structure V's nucleation, growth, and stability in the polymorphic system, which is so close and easy to transform each other, has ...
 GMP Manufacturing
Crystal Pharmatech offers a full spectrum of formulation manufacturing services covering a wide variety of dosage forms and meeting GMP requirements in the US, Europe and China.
GMP Manufacturing
Crystal Pharmatech offers a full spectrum of formulation manufacturing services covering a wide variety of dosage forms and meeting GMP requirements in the US, Europe and China.
 Amorphous Solid Dispersions: Two Heads Are Better Than One
Speaker: Dr. Na Li, Assistant Professor in Pharmaceutical Sciences at the University of Connecticut
Amorphous Solid Dispersions: Two Heads Are Better Than One
Speaker: Dr. Na Li, Assistant Professor in Pharmaceutical Sciences at the University of Connecticut  FIH-PMF-FMF Manufacturing
First-in-human (FIH) formulation development is done for phase I-IIa CTM Manufacturing to advance FIH clinical trial and obtain proof of concept (POC) of the safety and efficacy of the molecule under ...
FIH-PMF-FMF Manufacturing
First-in-human (FIH) formulation development is done for phase I-IIa CTM Manufacturing to advance FIH clinical trial and obtain proof of concept (POC) of the safety and efficacy of the molecule under ...
 The Bioprocessing Summit 2024 - Meet Crystal Bio at Booth T13
Join Crystal Bio's Co-founder, CTO, and Head of USA BD Ye Gu, Ph.D., as she presents at the Immuno-Oncology Summit 2024.Title: Analytical Methods to Assess Critical Quality Attribute in mRNA-LNP ...
The Bioprocessing Summit 2024 - Meet Crystal Bio at Booth T13
Join Crystal Bio's Co-founder, CTO, and Head of USA BD Ye Gu, Ph.D., as she presents at the Immuno-Oncology Summit 2024.Title: Analytical Methods to Assess Critical Quality Attribute in mRNA-LNP ...
 Contact Us
Contact Us
 Crystal Pharmatech and Particle Sciences Form a US-China Pharmaceutical CRO Partnership
By Damain GardeFrom Fierce BiotechPRINCETON, N.J. & SUZHOU, China--Crystal Pharmatech, a technology-driven solid-state research contract research organization (CRO) based in Suzhou, China, announc...
Crystal Pharmatech and Particle Sciences Form a US-China Pharmaceutical CRO Partnership
By Damain GardeFrom Fierce BiotechPRINCETON, N.J. & SUZHOU, China--Crystal Pharmatech, a technology-driven solid-state research contract research organization (CRO) based in Suzhou, China, announc...


